
Living on early seasteads will likely require living in close quarters with other people. And hot, damp environments are ideal breeding grounds for pathogens. Under such conditions, pathogens could rapidly infect everyone on a seastead. Maintaining good hygiene will be key to remaining in good health.
Liquid chlorine bleach (active ingredient: sodium hypochlorite) is one of the most effective, versatile, and cost-effective disinfecting solutions. It can be used to disinfect everything from surgical instruments to toilets to mold covered ship bilges. A year’s supply of sodium hypochlorite powder can be stored in a single 1 liter Nalgene bottle.
When used properly, bleach kills all of the following pathogens:
Bacteria:
Escherichia coli (E. coli)
Salmonella species
Listeria monocytogenes
Staphylococcus aureus
Clostridium difficile (C. diff)
Pseudomonas aeruginosa
Viruses:
Influenza (Flu virus)
Human Immunodeficiency Virus (HIV)
Hepatitis B and C viruses
Norovirus
Rhinovirus (common cold)
Adenovirus
Coronavirus (including SARS-CoV-2)
Fungi:
Candida species (e.g., Candida albicans)
Aspergillus species
Parasites:
Giardia
Cryptosporidium (although it may require higher concentrations or extended contact time)
Bleach directly destroys the cell walls of the pathogens, by breaking apart the carbon bonds (a chemical process known as "oxidation"). As a result, pathogens cannot easily evolve a defense against bleach like they can against antibiotic / antiviral drugs.
Here is the overall process for disinfecting a hard surface:
Dilute bleach to reach the desired concentration.
Wash the surface with hot, soapy water.
Rinse with fresh water.
Apply bleach solution to the entire area.
Leave the surface wet for at least 5 minutes.
Allow it to air dry or wipe dry with clean towels.
Chlorine loses its effectiveness quickly in the presence of oil, dirt and organic material, so replace the water with fresh bleach solution if it becomes dirty.
In the US, bleach is most commonly available in liquid form at most supermarkets. The active ingredient is typically sodium hypochlorite. For example, Clorox disinfecting bleach contains 7.5 grams of sodium hypochlorite dissolved in every 100 mL of fluid.
When sodium hypochlorite dissolves in water, hypochlorite (OCl) ions, and hypochlorous acid (HOCl) form and exist in an equilibrium with each other. Hypochlorous acid aka "free chlorine" is the primary active sanitizing agent, and is typically measured in parts per million (ppm).
To ensure bacterial/viral destruction, you must expose the pathogens to a 2000 ppm bleach solution for at least 5 minutes. Liquid bleach is typically sold at a much higher concentration than this, so you will typically want to dilute the bleach to reach the desired concentration. (Higher concentration solutions will also destroy pathogens, but the higher the concentration, the higher the risk that the bleach will damage the surface, harm a worker, or harm the patient/customer.)
According to the EPA, a sanitizing bleach solution contains a high enough concentration of bleach to kill bacteria (such as E. Coli), but does not necessarily kill viruses (such as Covid 19).
A disinfecting bleach solution contains a high enough concentration of bleach to kill both bacteria and viruses.
A typical sanitizing solution contains bleach at a concentration of 100 ppm. A typical disinfecting solution must contain at least 2000 ppm of sodium hypochlorite.
This table lists the amount of 7.5% bleach solution required to reach 2000 ppm in containers of various sizes.
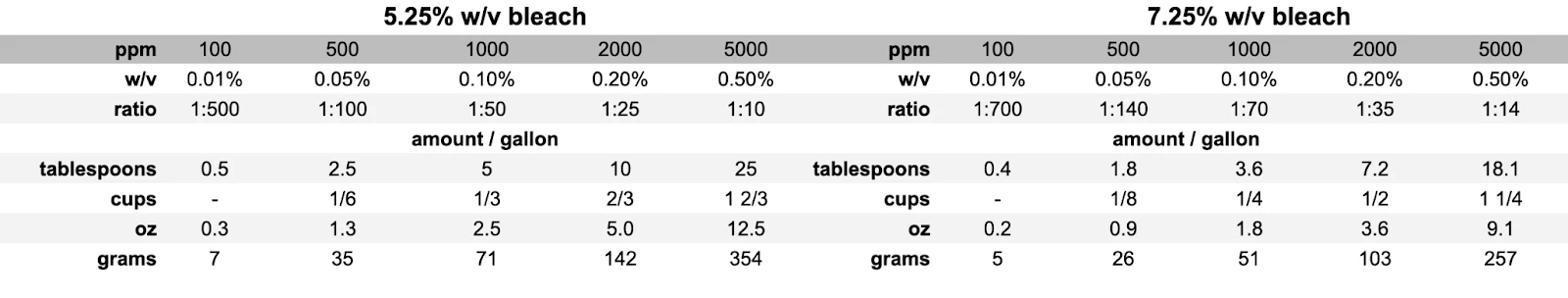
After rinsing, the residual bleach should be no higher than 0.5 ppm to avoid unpleasant tastes or smells. For example, typical municipal water systems produce potable water with a residual available chlorine level of 0.25 to 2 ppm.
Other vendors of liquid bleach might have higher or lower concentrations of sodium hypochlorite. You will need to adjust the concentration of bleach if you use a different formulation.
You can measure the chlorine concentration level with chlorine test strips with a measuring range of 0-2000 ppm or higher.
Warning!
DO NOT MIX BLEACH WITH OTHER CLEANING AGENTS!
When mixed with a number of other cleaning agents, bleach will form toxic/corrosive gases. Only mix bleach with plain dish soap / laundry detergent and water.
For example, when sodium hypochlorite mixes with ammonia, it will generate chloramine gas. Chloramine gas can irritate the lungs, and may cause fatal swelling ("chemical pneumonitis").
When bleach mixes with acids (vinegar, hydrochloric acid, etc) it will form toxic chlorine gas.
Bleach is also highly corrosive. Bleach fumes will rapidly corrode any nearby metals. Bleach solutions may irritate the skin and the fumes may irritate the respiratory tract. Highly concentrated bleach solutions should only be handled with adequate ventilation and protective gear (nitrile gloves, goggles, gas mask).
Chlorine bleach will also destroy the dye in many fabrics, so only wear clothing that you can afford to permanently bleach.
Buy only plain, unscented bleach, especially if you're using it to purify drinking water. Perfumes will make water taste terrible and may be toxic. "No splash" bleach contains gelling agents that will leave a film on surfaces.
Liquid bleach degrades over time. Sodium hypochlorite (3NaOCl) breaks down into salt (2NaCl) and sodium chlorate (NaClO3) at a rate of about 20 percent per year.
All of the following will also accelerate the rate at which bleach degrades:
Heat
Light
Humidity
As the concentration of free chlorine declines, bleach loses its effectiveness as a disinfecting agent. For consistent, maximum strength,
every 4-7 days from high test powder ( either > 70% sodium hypochlorite or > 70% calcium hypochlorite / pool shock).That said, bleach that has lost some of its potency can still be used–you just have to increase the concentration to reach a final dilution of 2000 ppm.
To reduce the rate of degradation, bleach should be kept in airtight, opaque plastic containers and stored in a cool, dark, dry place, away from any metal objects. The ideal storage temperature for bleach is between 50 and 70 degrees F. Whenever you buy bleach, write the date on the bottle with a Sharpie marker. Then try to use the oldest bleach first ("first-in, first-out").
You can also determine when your bleach was made by looking for the production code on the bottle.

For instance, in the example image above, A82223411 is the production code:
A8 refers to the plant that manufactured this particular bottle
22 represents the year 2022
234 is the 234th day of 2022 (August 22).
11 is the manufacturing shift identification code.
(31CA3 is the Environmental Protection Agency (EPA) registration code + state identification code.)
By following the above guidelines you can remain clean and healthy in a marine environment.
